Strados Labs Receives FDA 510(k) Clearance For Its Wireless Lung Sound Measurement Platform, RESP™
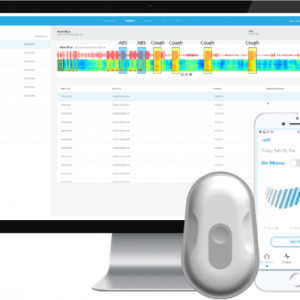
Strados Labs, a health technology developer of remote respiratory devices and software, today announced it has received FDA Class II 510(k) clearance for its first product, RESP™, a system for measuring, recording and analyzing lung sounds remotely across healthcare settings.
The RESP™ system has been used in clinical trials to help researchers quantify changes in lung sounds over time to compare against treatments, patient-reported outcomes, and other vital signs. The RESP™ can capture, store, and make available for clinical analysis these key changes in a patient’s lung sounds remotely with a small body-worn sensor and a cloud-based, HIPAA compliant software system. Dr. Mitchell Glass, the Chief Medical Officer of Strados Labs, said, “This wearable technology will allow us to follow our patients in healthcare settings more effectively, by providing regular interval listening between clinician visits, by archiving the patient’s lung sounds for future comparisons and by reducing the variability in auscultatory documentation that presents a major problem both in patient care and in clinical trials.”
Read the full article here.